
In the platform of Mass communication, we converted raw materials into finish products through finding the right news to cover for the media, using our voice in a responsible way to deliver the news, having the discernment if the news might affect, hurt, or wrong should not be shared to others but it should be right, would not hurt the people emotionally and should benefit them, and lastly we report not just to talk and share what we know but convert our knowledge to action, to act and be a part of the solution to the rapid negative changes in our society. The gram-to-mole and mole-to-gram conversion in particular is used constantly in both academic and real worl chemistry and biology. 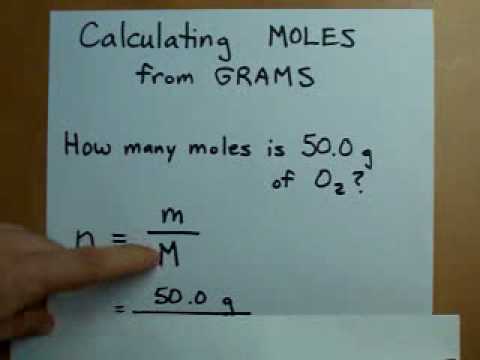
It can be applied to limiting reagent problems, concetration problems and much more. This lecture pertains in giving us the ability to do conversions with grams, moles and number of atoms/molecules is an essential skill for chemistry and life science students. The mole road can be very useful in learning how to convert back and forth among grams, moles, and number. The mole is at the center of any calculation involving amount of a substance. The gram-to-mole and mole-to-gram conversion in particular is used constantly in both academic and real world chemistry and biology. These types of conversions come up again and again throughout typical General Chemistry sequences and can be applied to limiting reagent problems, concentration problems, and much more. It also shows the conversion factors needed to interconvert between mass, number of particles, and volume of a gas. The Mole Road shows three quantities, mass, moles, and number of atoms or molecules, and how they are related to each other by atomic/molecular weight and Avogadro’s Number. I take you through converting step-by-step with discussion and examples!Ĭhella P. 
Having trouble with conversions? Perhaps you're taking a General Chemistry class in college. Here is an illustration of how to find moles when given the mass in grams of a substance: The mass of one mole of lithium. Conversions for Science Courses: Temperature, Conversion Factors, and the Metric System The grams to moles equation allows the simple conversion of grams to moles.Don't know how to find one? Don't worry! I'll show you how to calculate this value here with some review material and a worked-out example! Molecular weights are needed for all kinds of chemistry problems.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
